UPenn engineers are also developing chewing gum that destroys biofilm and other cool dental innovations.
Hyun “Michel” Koo grew up in what he calls a “poor section” of Sao Paulo, Brazil, witnessing—and sometimes experiencing—what happens when people can’t get oral health care.
Children suffering painful cavities and adults ashamed of missing teeth sparked a lifelong passion to make dental care more available and affordable for people in poor communities and countries. Now, as co-director of a groundbreaking research center at the University of Pennsylvania, where he is a dental school professor, he believes he is in position, along with like-minded and enthusiastic colleagues, to start making a dent in this abiding ambition as they develop innovative technologies with the potential to help transform oral health. They are asking things like:
What if, for example, inexpensive chewing gum could prevent plaque buildup, tooth decay, and gum disease? Or deliver antivirals against coronavirus?
What if swarms of micro-robots could substitute for brushing and flossing?
What else could smart technology and engineering do for problems like implant infections, cleft palates, and other dental troubles?
Meeting Koo in person for the first time in a large, windowless lecture hall at the Penn dental school in Philadelphia, his excitement was even more clear than in an earlier Zoom interview. The real-life occasion, on June 2, was the start of the inaugural scientific symposium of the Center for Innovation & Precision Dentistry (CiPD), where he had initially served as co-founding director and now serves as co-director along with the equally energetic and optimistic engineering professor Kathleen Stebe.
Comprising more than 20 members of UPenn’s Dental Medicine and Engineering and Applied Science faculties, this alliance of the schools, according to Koo, is thought to be the first university research center specifically designed to unite oral health clinicians and scientists with engineers in a formal partnership dedicated to rapidly and radically advancing oral health treatment and knowledge. The center began with a serendipitous research collaboration between Koo, Stebe, and others, which in turn led to a 2018 workshop funded by the university’s vice provost for research. Participants soon formed a task force that evolved into the core group of the new center. Though members had already published journal articles on some of the center’s projects before the symposium, the event was the center’s first chance to present a multi-faceted view of their work to the scientific world.
Dentists and engineers have obviously collaborated on many occasions on a wide range of treatments and devices, such as 3D printing of Invisalign orthodontic prostheses, using Dianodent lasers and sound pulses to find cavities earlier than customary methods can, or coating dental implants with antimicrobials, and doing minimally invasive robotic surgery to implant them in the first place.

Koo and his colleagues believe, however, that their center can supercharge progress by providing institutional, organizational, and financial backing, and soon, Koo hopes, a dedicated building that he expects will boost creativity by encouraging and facilitating the kind of informal, spontaneous interaction among colleagues from a variety of disciplines and academic statuses that makes ideas and collaborations sprout and grow. He also emphasizes the Center for Innovation & Precision Dentistry’s focus on finding solutions that are cost-effective and adaptable to the needs of people in under-resourced communities and countries—something largely missing from innovation in the nearly $39 billion global dental market, which generally directs its efforts toward equipping just the sort of dental offices and high-cost care that the world’s poor and marginalized cannot access.
Thanks to the center’s work, oral health experts are already learning about the many tools and resources that engineers can bring to analyzing and solving problems, and engineers are learning about the pressing and prominent oral health problems, says Kathleen Stebe. She and Koo intend to expand the cross fertilization among fields in other ways, too.
Last year, just after the Center for Innovation & Precision Dentistry came into existence, the National Institute of Dental and Craniofacial Research, one of the National Institutes of Health, recognized their potential by awarding them a five-year, $2.5 million training grant to prepare a cadre of postdoctoral researchers drawn from dentistry and oral health, several kinds of engineering, and other basic sciences including data science and computation, all dedicated to working at the intersection of oral health and high technology. Koo and Stebe believe universities and industry will both welcome the broad outlooks and expertise that these researchers will bring.
On this particular day, full of talks and a crowded poster session, five such innovations at the crossroads of dental health and technology stood out. The first of them illustrates a principle of useful innovation: acting “where the shoe pinches—solving the problem that needs to be solved,” said Penn dental professor Eugene Ko. He went on to explain how he discovered just such a need when close friends told him over dinner of their struggle to feed their new baby, who was born with a cleft palate. Among the most common birth defects, affecting about one in 1,700 newborns, the open spaces in the roof of the mouth and/or the upper lip, known as oral clefts, occur when tissues do not properly join together during prenatal development. The gaps prevent the baby’s mouth from forming the suction needed to suck nourishment from either breast or regular bottles. Special bottles currently available for infants with cleft palates are expensive, difficult to find, and hard to use. Ko immediately recognized a problem he thought he could solve.
Teaming up with Shu Yang, chair of the department of materials science and engineering, they explored several approaches, ultimately developing a one-way valve that traps milk in the chamber of a special nipple which allows the baby to extract it by biting rather than sucking. A specially designed screw collar attaches the modified nipple to regular nursing bottles. Ko is now currently working to test and commercialize the product.
LEDs, lasers, and piezoelectrics
A second project came from Geelsu Hwang, a dental school faculty member and core member of the Center for Innovation & Precision Dentistry. Hwang explained how a combination of laser technology and biochemistry could solve a big problem affecting one of the major oral health advances of the past several decades: the tendency of dental implants to fail because of infection or gum disease. Used today by more than 3 million people in the United States alone, implants replace missing teeth far more securely, comfortably, and durably than traditional dentures or bridges. But they require an invasive, expensive process of multiple oral surgeries over several months. In far too many cases, implants must be removed well before their expected decades-long useful lifespan, requiring more invasive and expensive procedures. Two important obstacles to success are that currently available implants are inactive and that users do not necessarily apply proper hygiene.
Specially engineered chewing gum can deliver pharmaceuticals to the mouth, including enzymes that destroy biofilm.
Hwang and colleagues have united a pair of advanced technologies into a “smart” implant that they believe can overcome these obstacles and provide healthy, long-lasting results. Trained in chemical and biomolecular engineering, Hwang has focused much of his research on biofilms, the sticky, bacteria-laden substance that clings to teeth and fosters oral disease. The new implant uses two dynamic technologies to allow the implant to defend itself.
First is photomodulation therapy, which uses laser light to make tissue, including the gum tissue surrounding an implant, “much more resistant to bacterial invasion,” Hwang explained. The second is piezoelectricity, a process in which certain materials produce an electrical charge when they are subjected to mechanical stress. The smart implant’s crown contains specially designed LED lights that provide infection-fighting phototherapy to the surrounding tissue, with the correct amount of light programmed into the device. “Ninety minutes exposure daily is the best,” Hwang says. Chewing and other natural mouth movements provide the mechanical stress needed to produce the electricity that powers the light. With a patent application under way, he added, the search is on for financing to allow a startup company to undertake the testing and clinical trials that could bring the device to market.
Smruti Nair, who is studying for her DScD degree, told me of the “revolutionary” work going on in the lab of her mentor, biochemist Henry Daniell, another core member of the Center for Innovation & Precision Dentistry. The lab’s goal, she explained, has long been to produce affordable drugs to end global health inequities and a low-cost and effective way of delivering them.
Daniell, along with biomolecular engineer Daeyeon Lee and other colleagues, are working to accomplish this with specially engineered chewing gum that can deliver a variety of inexpensively produced pharmaceuticals to the mouth, including enzymes that destroy biofilm. Removing this microbe-laden substance is an obvious necessity for oral health, but also a challenging problem because biofilms contain a matrix composed of biological polymers that protect the microbes and resists removal. An everyday toothbrush, the stick-with-bristles implement we generally use for this task, is an ancient technology that “goes back to the Egyptians… unchanged for millennia,” Koo says. Beyond that, he claims it is not particularly effective—nor is the periodic mechanical scraping we get at a dentist’s office. And equally important, a brush is useless for people with disabilities that prevent them from manipulating it.
Something to chew on
Daniell produces biofilm-fighting enzymes, among numerous other potentially useful proteins, with a patented, novel process that grows them in plants. Bombarding them with the DNA of the desired protein causes the chloroplasts, which are intercellular elements of all green plants, to take up the genetic material and produce the desired molecules in their leaves. Once the plants are grown, the molecules of interest are extracted, freeze-dried, and ground up, creating powder ready to be packed into a pill or added to a gum.
But to be effective against biofilm, the enzymes must be protected on the way past the mouth’s salivary defenses, which ordinarily can break them down. That’s where the special gum—which has been engineered with features that keep the enzymes intact—comes in.
Swarms of micro robots could be used to clear away biofilm swiftly, safely, and conveniently.
In another promising use, delivering a different enzyme, ACE2, via the gum shows great promise in reducing transmission and preventing infection by the Sars-CoV-2 coronavirus, Nair noted. The FDA has recently given it the go-ahead for clinical trials.
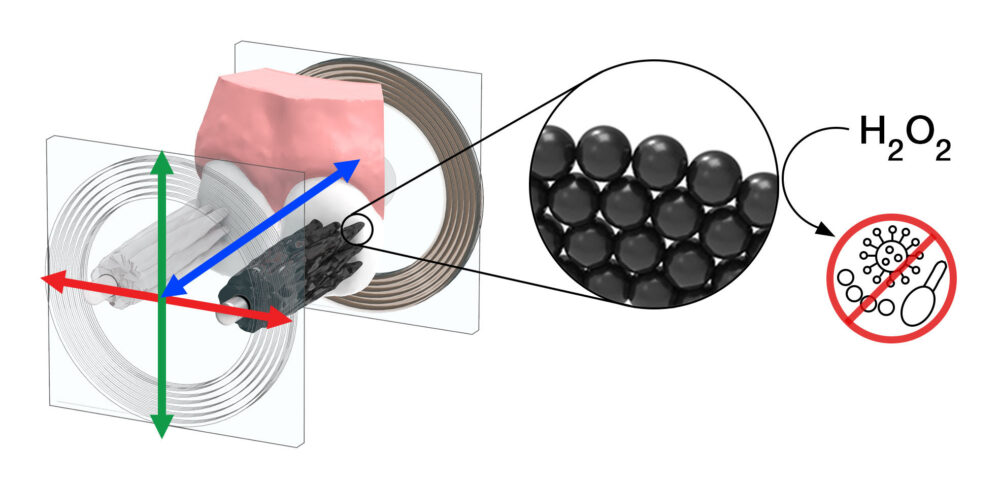
Koo had spent years researching biofilms and was seeking a new way to clean them from the teeth, searching for a method that would meet his criteria: It had to be practical, scalable, and low-cost. He and colleagues had experimented with nanoparticles that contain iron oxide which, through catalytic action, can cause hydrogen peroxide to release free radicals that can break up biofilms. Through a friend he learned of work in the engineering school with a very similar kind of iron oxide-containing nanoparticles. The engineers, however, were turning them into micro-robots by using magnetic fields to guide them in specific movements. What about guiding them across, around, and among teeth? Joining forces across disciplines, Koo, Steager, Stebe, and others envisioned how swarms of microrobots could be used to clear away biofilm swiftly, safely, and conveniently.
Together, the groups devised two different systems for what they call catalytic antimicrobial robots to break down and remove biofilms. The first approach suspends nanoparticles in a solution and the second embeds them in gels molded into various three-dimensional forms. The nanoparticles clear biofilms from surfaces like the front of your teeth, much as a snow plow clears snow, and also from enclosed spaces and narrow passages, such as tight spaces between teeth.
Tests using flat panes of glass and glass tubes showed that both approaches can precisely hone in on and destroy the bacterial biofilms and matrix, and clear away the debris so completely that the biofilms have little chance of growing back.
And as a bonus, Koo says, the FDA-approved nanoparticles, which “cost a couple of cents a gram,” can also be used to treat iron deficiency anemia, another health problem that often affects poor and malnourished populations. Thus one solution can address two big problems, he says.
For all the ideas and projects explored at the symposium, Penn researchers are hardly the only ones working at the frontier of innovation in oral health science and engineering. To name just two examples, the American Dental Association’s Science & Research Institute in Gaithersburg, MD, works to develop and help commercialize innovative solutions to oral health problems and Harvard bioengineer David Mooney is working to develop biomaterials to aid in surgery and cell regeneration. But the center, with its leadership, integration, and sense of mission, seems well positioned to be a major player.
Editor’s note: This story were updated on August 19, 2022, to reflect that Koo is the co-founder of the CiPD center and to clarify his statement in the second to last paragraph.













