A new wave of spit-and-swab home testing kits promises to give individuals more control over their own health.
I started to feel sick on my way home from vacation in early July. At first, I thought my muscles were sore from a five-mile hike the day before. Then came a low-grade fever, chills, burning eyes, and exhaustion so deep, I didn’t have the energy to even think about driving to a doctor’s office.
Two and a half years into the pandemic, I assumed I had COVID-19, and I confirmed the diagnosis as soon as I stumbled into my kitchen with a free antigen test that turned positive instantly. After retreating to my bedroom with Tylenol, a thermometer, an air purifier, and orange juice, I tested several more times over the next couple of weeks. Rapid tests remained positive on days 5, 7, and 9, when a faint line appeared after a long while.
A negative result on day 11 felt like a triumph—not just of wellness returning but also of the promise of modern diagnostics, which gave me the ability to figure out for myself what was infecting me without ever having to sit in a clinic waiting room, where I might get other people sick, pay out of pocket for the privilege, and wait days for the results.
We may be on the precipice of a new era in health care that depends on self-testing for a growing number of health issues.
With the active development and widespread distribution of at-home tests for COVID-19, along with accumulating evidence that self-tests can be reliable and useful, we may be on the precipice of a new era in health care that depends on self-testing for a growing number of health issues, particularly for infectious diseases, experts say. By the yardstick of the pandemic, that era is already here.
The global self-testing market was valued at nearly $6 billion in 2020, according to a report by the market research firm Brandessence. That market is projected to grow to $8 billion by 2027, and numerous products are under development by many major pharmaceutical brands, including Johnson & Johnson, Roche, Abbott, and Quest Diagnostics. Applications include celiac disease, thyroid disease, allergies, menopause, vitamin levels, sexually transmitted diseases, and much more.
But the future of self-testing is also filled with obstacles, risks, and caveats. For a new test to come to market, companies must have a financial incentive to develop it, which means they need to be convinced consumers will buy it. Even when companies make tests, getting approval from the U.S. Food and Drug Administration (FDA) requires a grueling regulatory process. And to be useful—and beneficial—from a public health perspective, people should be able to make good decisions about which tests to take and what to do with the information they get, says Barry Lutz, a bioengineer at the University of Washington in Seattle and one of multiple principal investigators on the Seattle Flu Study, which was investigating the value of home testing for influenza before COVID-19 came along.
After years of slow progress, the pandemic may have finally broken down some of those barriers to development. “COVID-19 has completely changed the landscape,” Lutz says. “It forced the FDA to really consider home testing. At this point, with that groundwork being done and that gate being open, I expect that other tests that are available in the clinic now will start to find a way into the home.”
Before the pandemic
The technology behind rapid testing for COVID-19 has been available for at least 15 years, says William Morice, chair of the department of laboratory medicine and pathology at the Mayo Clinic in Rochester, Minnesota. The concept is straightforward and similar to the way home pregnancy tests work. But as with home pregnancy tests, it has historically been extremely difficult to get the FDA to approve rapid tests for home use, even when they were cleared for use in clinical settings. The agency questioned whether ordinary people could test themselves without making mistakes. Providers, too, were hesitant to accept home testing results because of both safety and liability concerns. If people were going to have to pay out of pocket for self-tests and then go to a doctor to verify their results anyway, what was the point of testing at home in the first place?
Research on influenza, which began before the pandemic, started to show that people can use home tests. In a 2019 study that monitored 481 people in 124 families, rapid flu tests detected about 70 percent of influenza cases, not as good as PCR tests but similar to the sensitivity of rapid tests done in clinical settings. Still, when their research on self-testing for influenza started, Lutz says, it was expected to be a slow process, and they were hoping to push the field forward incrementally. He didn’t know of any submissions for FDA-approval for at-home influenza tests at the time.
The pandemic changed all that. Ever since the agency started to give emergency-use authorization to rapid COVID-19 tests in November 2020, multiple companies have submitted applications for at-home influenza test kits.
“Now that people are used to testing and providers have started to open up to being willing to accept these results, insurance companies will eventually see the value in this and pay for it,” Lutz says. “COVID-19 just accelerated everything.”
Tracing back to the HIV pandemic
A look to the past illustrates just how quickly COVID-19 changed the industry landscape for self-testing. The first—and before COVID-19, the only—government-approved home test kit for an infectious disease was an HIV test the FDA authorized in 1996, which allowed people to prick their fingers at home and send samples of blood for analysis in a lab. In 2012, the agency approved a less painful self-test for HIV that uses cheek swabs to collect saliva and delivers results directly to users in 20 to 40 minutes.
According to historical accounts, both approvals reignited controversies that began in the 1980s about whether empowerment and destigmatization through self-testing initiatives would outweigh the potential for missed infections and psychological reactions to testing positive outside of the health care settings, where treatment could be initiated immediately and counseling and information were available.
Nitika Pant Pai, a physician scientist and epidemiologist at McGill University in Montreal, became interested in HIV self-testing about 15 years ago, after conducting a project in rural India that trained health care workers to test pregnant women in labor for HIV. The goal was to link women with HIV to care so they could be immediately treated with antiretroviral therapy to improve their health outcomes and prevent them from transmitting the virus to their babies.
During that project, Pant Pai says, her patients started to ask her if they could take the tests to do at home, which wasn’t allowed at the time in India or Canada. But when she looked into it, institutional review boards (IRBs) told her that giving people the ability to find out if they were infected would increase suicide rates—the same argument once used to deny at-home pregnancy testing. In addition, “The IRB said, ‘Oh, we already have these rapid tests for HIV. We don’t need anything called a self-test,’” Pant Pai says. “The concept was a little far-fetched, even in HIV.”
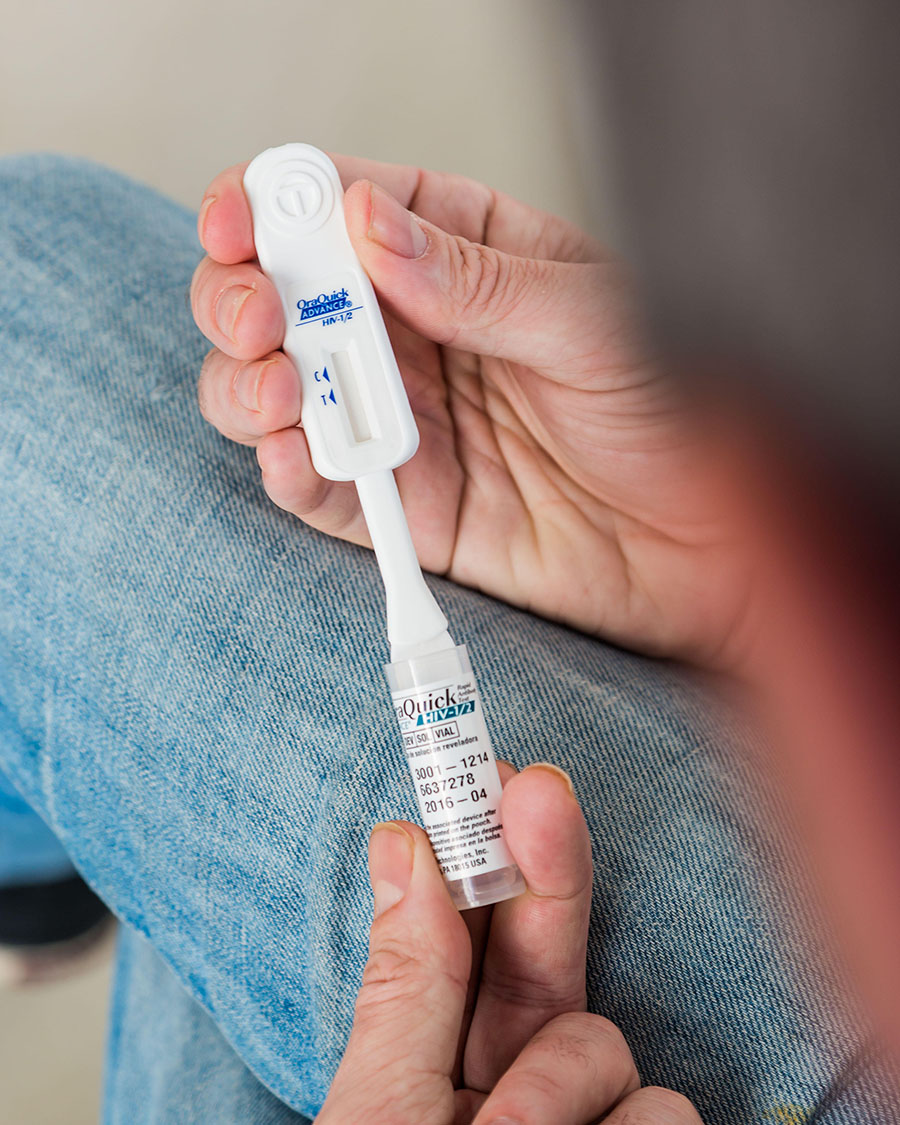
Since then, evidence has accumulated to show that people can test themselves correctly for HIV, that self-tests are accurate enough to be useful, that people can handle the information, and that having the option of self-testing encourages people to get tested who might not otherwise do it. In the last two years, as part of a recent effort to increase screening rates among people at high risk of HIV in underserved areas, including gay and bisexual men in Black and Hispanic communities, the U.S. Centers for Disease Control and Prevention (CDC) reached out to people around the country through dating apps, among other strategies. This allowed CDC researchers to inform people about a website that allowed them to privately order at-home HIV tests. Between February and October 2021, more than 56,000 people ordered tests, researchers reported in 2022 at a virtual conference on retroviruses and opportunistic infections. More than 25 percent of those who responded to a survey said they had never been tested for HIV before.
Sexually transmitted diseases remain a major target for self-testing because of ongoing stigma that makes testing in private appealing, experts say. Multiple at-home tests for hepatitis C exist, though they require people to either use a device that collects blood directly through the skin or take a finger-prick and leave a dab of blood on paper that they send to the company for analysis. Self-sampling swabs that people mail in for results are also FDA-approved for chlamydia, gonorrhea, and human papilloma virus, Pant Pai says. These sorts of tests were particularly useful at the beginning of the coronavirus pandemic, according to a 2022 analysis from Johns Hopkins, because most STD clinics stopped offering in-person services. But even after the initial shutdowns were over, orders surged for home-based STD test kits as a result of increased referrals from health departments, according to the researchers.
The availability of at-home testing for sexually transmitted infections may even be transforming the way people date. In 2021, a British company called iPlaySafe released an app which allows users to send in samples and then share their results as part of their dating profiles. The company’s product tests blood and urine samples for a panel of sexually transmitted diseases, including chlamydia, gonorrhea, HIV, syphilis, hepatitis B, and hepatitis C. Bacterial vaginosis, which is not an STD but is linked to sexual activity, can also be tested for at home, requiring a pH level vaginal swab that quickly turns color in response to a bacterial or yeast infection.
“If you look at what COVID-19 did for self-testing and for home testing, I think COVID-19 has been a blessing.”
From a consumer standpoint, the desire to monitor what’s happening internally extends beyond infectious diseases. That interest has fed a burgeoning market of products that cater to data-driven biohacker types. Some devices currently available allow users to get detailed information about vitamin levels relevant to athletic performance. Others gauge women’s health markers, including thyroid levels and diabetes risks. And we may be just at the beginning of the trend. Home testing for a broad range of health indicators on demand could be part of the push for individually tailored precision medicine, according to some companies that offer nutrition and fitness advice based on daily or weekly monitoring done at home.
COVID-19 helped show the world what self-testing is and what it can do, Pant Pai says, offering an alternative to what she sees as a patriarchal model of health care that resists giving individuals control. “If you look at what COVID-19 did for self-testing and for home testing, I think COVID-19 has been a blessing,” she says. “Testing was always something that you go to a doctor and get. But now I think COVID-19 has brought about that shift that you can do it in the comfort of your home, and that would not have happened if [the pandemic] did not happen.”
Barriers at home
Despite the promise of home-testing for infectious diseases, plenty of obstacles remain, including scientific challenges, Lutz says. Making lateral-flow antigen tests as accurate as possible depends on finding a useful antibody to attach to the test strip, and some antibodies work better than others. The stability of the viral protein also varies. Rapid influenza tests may only detect about 50 or 60 percent of infections, Morice says, depending on things like how much virus a person is shedding, their particular strain of influenza, and variations in sample collecting. Translating that uncertainty into an action plan is hard enough to do in a clinic, particularly when there are more accurate tests available. “It’s like flipping a coin,” he says. “What are you going to do with it?”
To make tests more accurate, some researchers are experimenting with molecules called aptamers, which are essentially fragments of DNA or RNA that take the shape of antibodies and could make rapid tests both cheaper and more programmable to detect specific viruses, Lutz says. In May, the Swiss company Achiko announced they had developed a COVID-19 rapid test that uses aptamers to achieve a sensitivity rate of 97 percent, more sensitive than antibody-based products.
Work is also ongoing to make results easier to see with fluorescence, Lutz says. And some researchers are trying to reduce the price of PCR-type tests. At least three COVID-19 PCR tests are already available under FDA Emergency Use Authorization, but they currently cost several times more than rapid antigen tests.
From an ethical perspective, experts are grappling with the potential for an exploding at-home testing market to exacerbate the kinds of concerns raised by widespread screening programs. Studies of programs to screen for breast cancer, prostate cancer, thyroid cancer, and others have found that testing large numbers of asymptomatic people can increase the rates of anxiety and invasive follow-up testing without always saving lives. “If you’re letting people choose which tests they’re going to run,” Lutz says, “they may be running tests when they don’t really need to run them.”
On the flip side, developers of tests for home diagnostics may face economic barriers that prevent them from reaching the marketplace, says David Simon, a law professor who studies drugs, devices, and liability at Harvard Law School. Companies have to consider whether there is a market for a test and whether people will be able to afford it, either out-of-pocket or through insurance. “Businesses are not going to develop tests if they can’t turn a profit,” Simon says. “It just doesn’t make any sense.”
Legal issues add to the complexity. Insurance companies are more likely to cover diagnostics or treatments that are FDA-approved, cleared, or authorized. But getting that approval can be an expensive and time-consuming process. Instead, some companies market diagnostic products in the “wellness” category, using disclaimers and marketing language that emphasize patient empowerment, Simon and colleagues wrote in a 2022 analysis. Once companies have built a customer base, they might eventually submit to the FDA to enable marketing their product as a device.
Regulatory procedures can be confusing, even for consumers who dig into government websites to figure it out. A quick search online or at your local drug store will turn up over-the-counter, at-home test kits for, among many others, urinary tract infections, influenza, strep, vitamin levels, allergies, and thyroid disease. There are apps and devices on the market that claim to diagnose seizure-like activity, atrial fibrillation, ADHD, Alzheimer’s disease, and more. But not all of these tests and products are considered by the FDA. For tests that are sold without a prescription but require sending a sample in to a lab, regulation goes through Clinical Laboratory Improvement Amendments (CLIA), which requires laboratory certification by the Centers for Medicare and Medicaid Services, and may also be subject to FDA regulation, but not always. The FDA also does not review all at-home direct-to-consumer tests, Simon says, depending on risks posed by the tests and whether they are intended for general wellness or diagnostic uses.
Drug companies have a challenging bet to make in uncertain markets, Morice says. For COVID-19, a surge in demand led to the quick development of rapid tests, but sales have since flagged. According to a CDC analysis of at-home testing use for the six months prior to March 2022, the highest rate of use was in January 2022, when 11 percent of people surveyed said they had tested themselves over the last 30 days—compared with 2 percent in October 2021. And although Abbott reported record sales in the first quarter of this year because of COVID-19 tests, the company has projected that sales will slow over the rest of the year.
At-home kits currently on or near the marketplace—A tiny sampling of what’s available
Questions remain about how many tests people will buy for COVID-19 and other conditions in the months ahead. “On the one hand, they don’t want to over-invest because maybe this is something of a mirage, and people are going to go back more to normal,” Morice says. “The flip side is they don’t want to get caught flat-footed if all of a sudden there’s a real surge and this becomes commonplace in health care.”
As companies work through the hurdles, the self-testing market is a competitive and global landscape of research and development that is new enough to be wide open, Lutz says. Targets include both high-income countries, where people want privacy and convenience, and low- and middle-income countries, where infrastructure gaps, staffing issues, stigma, and other factors can get in the way of diagnosing and treating infections, Morice says.
In addition to sexually transmitted diseases, Lutz says, prime candidates for infectious disease self-tests include respiratory viruses, which transmit easily and can be detected using already-available technologies. The list of available products may grow to include respiratory syncytial virus (RSV), whooping cough, and panels that test for multiple pathogens at once. The company BD Veritor is already selling a combined test for coronavirus and influenza that delivers results digitally in 15 minutes. Labcorp gained emergency-use approval this spring for a combined COVID-19, influenza, and RSV test that requires people to take samples at home and send them in for analysis.
The tests I did in my kitchen back in July didn’t just confirm why my body felt like it had been run over by a truck. They are likely just a taste of how I’ll deal with feeling awful in the future.
“There are lots of companies who have accelerated their development, and there are many new entrants who are developing diagnostics,” Lutz says. “There’s really a green field out there for the companies that can get it right.”









