Serimmune’s at-home tests compare your “epitope repertoire” against known cases, for more accurate diagnoses of everything from COVID-19 to Lyme disease to Epstein-Barr.
I’m writing this almost exactly three years after I first heard of the coronavirus. Although by now just about everyone I know has suffered a COVID-19 infection (or two), so far I’ve somehow escaped without so much as a sniffle. Am I off the hook? Or will I eventually succumb like everyone else? To find out, I’ve been participating in a clinical study from the Goleta, California-based biotech company, Serimmune. They hope to answer that question, not just for COVID-19 but for dozens and potentially, all infectious diseases.
Viral pathogens affect everyone differently, barely noticeable in some, but causing weeks of bedridden agony or worse in others. Whether you get sick or not can depend on your previous infection history, exposure level, general health factors like obesity, or (especially with COVID-19) age. We all know people who were infected despite doing everything “right,” and vice versa: those who appear the most reckless seem to get off scot-free. When proto.life contributor and long-time self-tracker Esther Dyson quantified her vaccination experience back in 2021, she suspected she had been infected as early as February 2020, though her subsequent antibody tests all showed negative. Did she really have it? Is she still at risk? Like her, I’m active, healthy, and vaccinated—but I sure would like to know if I’m out of the woods.
That pile of COVID-19 tests lying in your bathroom closet are based on the simple fact that antibodies are proteins, each with a unique shape that can bind with chemicals that change color when a match happens. Your at-home COVID-19 test binds with just one of the antigens associated with the SARS-CoV-2 virus, the receptor binding domain of the so-called “spike protein.” Even if that cotton swab you rubbed in your nose was smothered with live virus antigens, the test would show negative unless it’s got the one specific antigen to that spike protein. (Oh, and vice versa: as every TikTok-watching kid eager to skip school knows, a little Diet Coke on that strip will cause a false positive, as acidic liquids mess up the antibody-sensing proteins and make the nanoparticles under the T appear red)
Why can’t you design an antibody test that looks at all the possible antibodies rather than just one at a time?
The scientists at Serimmune have been studying antibodies for years, and they wondered why these tests have to be so specific. Why can’t you design an antibody test that looks at all the possible antibodies rather than just one at a time?
One problem is that your body is loaded with antibodies. Since birth, every time you were ever exposed to a pathogen, your immune system neutralized it with antibodies, most of which are in your body’s “repertoire” somewhere, ready to bounce back in case that same pathogen arrives again. You have literally millions of different antibodies, like a visitor log from every invasion attempted against you. Someday science may be able to catalog them all, but that day is far in the future.
The Serimmune scientists decided not to bother trying to identify each of them individually—there are too many. Instead, they use advanced tricks with bacteria and sequencing machines to break your sample into a digitally encoded “epitope repertoire,” a million unique pieces that represent antibody “keys” that fit into the “locks” found in pathogens. Whereas conventional tests for, say, Lyme disease antibodies, look just at the “locks” that scientists have painstakingly identified as matches for Lyme-causing microbes, Serimmune uses machine learning to identify how well your overall epitope repertoire resembles the repertoire of people with known Lyme infections. That’s a lot of information, and the pattern-matching involves some clever tricks to simplify the torrent of data, but ultimately the scientists claim they are able to match pretty well—even against a completely new disease like COVID-19.
The test uses a tiny blood sample, from an easy at-home kit the company has been sending me every six months (I’ve just finished my third one). I plop a special teacup-sized collection device on my upper arm, wait about 10 minutes for it to capture a few drops of blood, and send the package to their labs. A few weeks later I have my results.
Serimmune gives me a COVID-19 Total Antibody Score, their measure of the quantity and type of SARS-CoV-2 -related antibodies they found. On a score from 0 to 100 (it’s a normalized scale, where higher is “better”), I started at just under 80 about a year ago, which was a few weeks after my latest shot (it was my third, a Pfizer/BioNTech booster). But since then my antibody score has been steadily plummeting, down to 31 on my latest test (December 2022). I’m getting very close to pre-pandemic antibody levels. Is that bad?
Preliminary Results
Like most things in immunology, it depends. So far Serimmune has collected more than 3,000 samples from close to 2,000 people like me enrolled in their study—not to mention tens of thousands more in their database from other studies. While it’s far too early for Serimmune to release official results—no doubt in a peer-reviewed journal like Nature’s Communications Biology, which published some preliminary findings in late 2021—the company has been refreshingly transparent with the data they’ve collected in their clinical study so far, letting us view anonymized results like these:
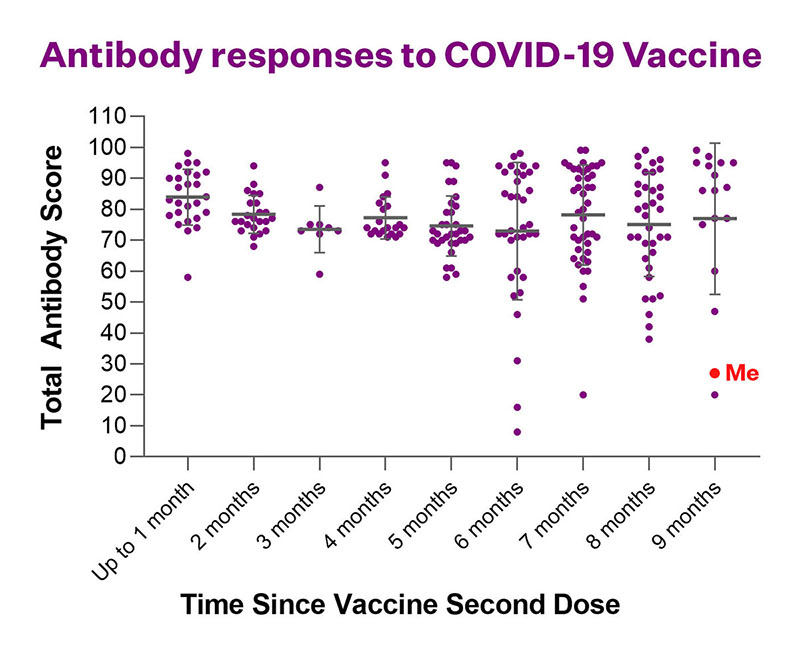
The news for me is frankly not good. While no test is perfect, it looks like as far as antibodies are concerned, my vaccine-initiated protection against COVID-19 has nearly faded since my booster more than a year ago.
Then why haven’t I been infected, like nearly everyone else around me? And given my apparently waning antibodies, should I get another booster, maybe one of those new bivalent ones? Here’s where science, like life, gets tricky. When, as of late 2022, Serimmune compared the hundreds of infections in their dataset, they found breakthrough infections were just as common among those, like me, who’d had a third shot versus those who’d had only two. While CDC and other trustworthy authorities point to data recommending additional shots to prevent worse outcomes, clearly, antibody levels alone aren’t the whole story.
Incidentally, Serimmune’s study includes more than a hundred cases of long COVID as well. Compared to infected individuals who didn’t report long COVID symptoms, in about a quarter of long COVID cases the antibody scores against the spike protein are dramatically lower than among infected people who fully recovered. In other words, the test seems to identify notable differences in some long COVID cases, providing hope for a more rigorous diagnosis and potential treatment.
Other uses for the technology
While (hopefully) the worst days of COVID-19 are behind us, there are plenty of other conditions where a general antibody test like this one could prove useful. For those study participants like me who opt into additional reports, Serimmune shows antibodies against several tick-borne diseases (Lyme, ehrlichiosis, etc.), the tropical parasite-caused Chagas disease, and a marker for celiac. It’s already made a difference in one of their study subjects, a woman whose many chronic ailments went undiagnosed for years because her lab tests always seemed normal. The Serimmune test, looking at her entire epitope repertoire, at last suggested a treatment option when it turned up positive for Lyme-related antibodies.
Even diseases that may have nothing to do with infections, like autoimmune conditions, allergies, and many cancers, generate antibodies that can be detected in a broad test like this one.
So will I get COVID-19 or not? I asked Serimmune’s senior director of R&D, Kathy Kamath. Finding that answer, not just for COVID-19 but for every other infectious disease is “the holy grail,” she says, but meanwhile we just don’t know. Their technology lets them easily compare the “epitope repertoire” of vaccinated people with breakthrough infections with those like me who haven’t yet had an infection.
Ultimately, none of us is really out of the woods from viral infections. We do what we can—vaccination is almost certainly what has protected me so far—but viruses and other pathogens have been a part of life since, well, the beginnings of life itself. Maybe by the time you read this, COVID-19 will at last have caught up with me, but even if it doesn’t, I’ll inevitably fall prey to other viruses throughout my lifetime. But in our never-ending fight against them, here’s hoping technologies like Serimmune can help all of us beat the odds.